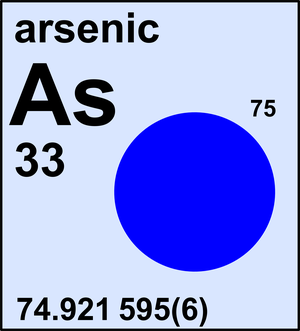
Arsenic
| Isotope | Atomic mass (Da) | Isotopic abundance (amount fraction) |
|---|---|---|
| 75As | 74.921 595(6) | 1 |
Arsenic is a monoisotopic element and its atomic weight is determined solely by its isotope 75As. The Commission last revised the standard atomic weight of arsenic in 2013 based on the latest Atomic Mass Evaluation by IUPAP.
© IUPAC 2003
CIAAW
Arsenic
Ar(As) = 74.921 595(6) since 2013
The name derives from the Latin arsenicium and the Greek arsenikos for "masculine" or "male" because
the ancients thought that metals were different sexes. Arsenic was known in prehistoric times for its poisonous
sulfides. The German scientist and philosopher, Albert von Bollstadt (Albert the Great or Albertus Magnus) is thought to have obtained the metal around 1250.


