Krypton
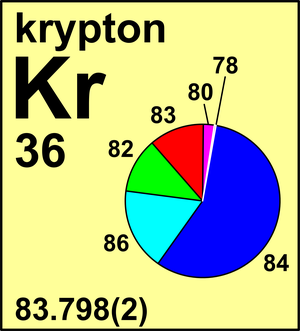
| Isotope | Atomic mass (Da) | Isotopic abundance (amount fraction) |
|---|---|---|
| 78Kr | 77.920 366(2) | 0.003 55(3) |
| 80Kr | 79.916 378(5) | 0.022 86(10) |
| 82Kr | 81.913 481 15(4) | 0.115 93(31) |
| 83Kr | 82.914 126 52(6) | 0.115 00(19) |
| 84Kr | 83.911 497 73(3) | 0.569 87(15) |
| 86Kr | 85.910 610 63(3) | 0.172 79(41) |
The atomic weight of krypton is based on analyses of krypton separated from air. In its 1961 report, the Commission recommended Ar(Kr) = 83.80 based on the isotope-abundance measurements by Nier. The standard atomic weight and uncertainty of krypton were changed to their current values in 2001 as a result of new mass-spectrometric measurements.
Recent reviews have concluded that the vast majority of krypton in and on the Earth is indistinguishable isotopically from atmospheric krypton. However, minor localized occurrences may be found that have been fractionated isotopically by processes such as diffusion or crystal-liquid partitioning. Also, the isotope abundances of krypton may be altered locally by spontaneous or neutron-induced fission of uranium, or by neutron capture by bromine.
The radioactive isotopes 81Kr (cosmogenic) and 85Kr (released from nuclear industry) are present in the atmosphere and hydrosphere and are used as environmental tracers in hydrology and oceanography, but their abundances are several orders of magnitude too small to have a measurable effect on the atomic weight of krypton.
© IUPAC 2003
CIAAW
Krypton
Ar(Kr) = 83.798(2) since 2001
The name derives from the Greek kryptos for "concealed" or "hidden". It was discovered in liquefied
atmospheric air by the Scottish chemist William Ramsay and the English chemist Morris William
Travers in 1898. A wavelength in the atomic spectrum of 86Kr is a fundamental standard of length.


